Development of four conceptually different New Plant Breeding Techniques
The objective of CHIC’s in this area is to develop four different methods for genome editing of chicory. These methods differ in their degree of ‘DNA invasiveness’: the extent to which CRISPR genes are introduced into the plant’s DNA. In the end all four methods lead to identical genetic outcomes.
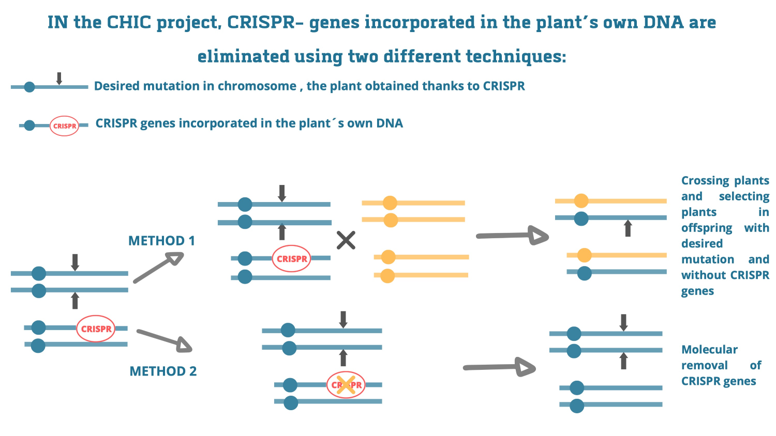
CHIC methods related to the presence of CRISPR DNA in the chicory plant’s DNA
These two methods are based on stable integration of CRISPR genes in the chicory genome, leading to so called transgenic chicory plants. After the CRISPR genes have done their work, the genes are removed from the genome. In the 1st method the crossing and selection will be used to select offspring plants without the CRISPR genes and with the desired mutation. In method 2, specialized enzymes will be used to remove the CRISPR genes from the plant’s DNA, so making crosses is not needed.
During the 1st two years of the project, thanks to applying the 1st method we have obtained transgenic chicory plants. These plants are now being analyzed for functional edits in the chosen target genes: genes involved in the production of the enzyme germacrene A synthase. This enzyme is essential for the production of terpenes.
Methods related to the non-presence of CRISPR genes in the plant’s DNA
These two methods are based on administering CRISPR tools to chicory cells without incorporating CRISPR genes in the plant’s DNA.
In method 3 plasmid-DNA that harbours CRISPR genes, is introduced into individual chicory cells. Plasmid DNA is best known from bacteria: circular DNA molecules that encode important traits. After introduction into a chicory cell, the CRISPR DNA is expressed and the CRISPR tools are being assembled in the cell. The plasmid DNA is eventually degraded.
In method 4 the CRISPR tools, protein and guiding RNA, are assembled outside the plant cell and then introduced into plant cells. This way there is no CRISPR DNA entering the plant cells.
For all methods we need to treat individual chicory cells (protoplast). The challenge then is to grow complete plants from these single cells. We have shown that this is indeed possible. We have also optimized techniques to introduce plasmid DNA and the CRISPR tools into chicory cells.
This has already resulted in plant cells in which the genes involved in the production of the enzyme germacrene A synthase are permanently disabled, thanks to the small alteration the CRISPR tools made in the plant DNA.
Optimizing the CRISPR tools
One of the CRISPR tools is a guide RNA (gRNA) molecule. The other is an enzyme called Cas9. The gRNA guides the Cas9 enzyme to the place in the plants’ DNA where a mutation is desired. We have designed gRNAs and have shown that they indeed guide the enzyme, enabling it to temporarily break the DNA at the correct place.
We have obtained plants with desired mutations in both chromosome sets (chicory is diploid) and in many or even all the gene copies chicory possesses. We are now testing the plants for the terpene content that may have some type of bioactivity.
Genome insight
The existing DNA screening techniques are not effective to screen plant material for small changes in the DNA in a high throughput manner. Therefore we are developing a new technique, based on techniques one of the partners developed for kiwi.
During 2019, the partners decided to jointly invest into a better assembled genome sequence of chicory, as the available genomic databases were of insufficient quality for our research.

 This project has received funding from the EU Horizon 2020 research & innovation programme under grant agreement N. 760891.
This project has received funding from the EU Horizon 2020 research & innovation programme under grant agreement N. 760891.